Prospective Breast Cancer Biobank
A population-based biobank in Western Norway with a long follow-up time.
The Prospective Breast Cancer Biobank (PBCB) is a population-based biobank in Western Norway with long follow-up. Breast cancer remains the most prevalent malignancy among women worldwide. Recent advances in biomedical research enable longitudinal monitoring of patients, facilitating evaluation of the long-term efficacy of current therapeutic strategies and the identification of predictive biomarkers for early detection of recurrence. PBCB provides systematic follow-up of breast cancer patients for up to 11 years, establishing a robust and unique platform for translational research and precision oncology within this patient population.
The full protocol is described in detail in Søiland H. et al., 2022 (e054404.full.pdf)
In Norway, over 40,000 women and around 30 men are diagnosed with breast cancer each year, and more than 60,000 women are currently living after undergoing treatment with varying degrees of side effects. Despite the generally good prognosis for this patient group, some will experience that the side effects reduce their quality of life. Additionally, there is a risk of recurrence of the disease. Therefore, there is a need to regularly follow up with patients to assess quality of life and to collect liquid biopsies such as blood or urine samples. The follow-up will indeed contribute to optimizing treatment, reducing side effects, and detecting recurrences at an early stage—so that new treatment can be initiated early.
PBCB has been collecting liquid biopsies in the form of blood and urine samples along with clinical information and patient-reported data (questionnaires/PROM) from 1,455 patients who have undergone surgery for breast cancer at Haukeland University Hospital (HUS) and Stavanger University Hospital (SUS) since 2012. This collection enables several important types of studies on breast cancer in our region and forms the basis for research collaboration both nationally and internationally.
Research Group at HUS
The PBCB research group at Haukeland University Hospital consists of researchers and clinicians from the hormone laboratory and the endocrine surgical department.
The hormone laboratory's research group is led by professors Gunnar Mellgren. The group researches breast cancer with a focus on endocrine treatment and advanced drug analysis (mass spectrometry). Additionally, the group works on several projects focusing on obesity, nutrition, and type 2 diabetes. The group is affiliated with the Mohn Nutrition Research Laboratory and Clinical Institute 2 at the University of Bergen.
https://www4.uib.no/en/research/research-groups/hormone-laboratory-research-group
The endocrine surgical department is led by Dr. med. Anette Heie, and the department has been central to the collection of patient samples and questionnaires for PBCB. Research projects have been conducted at this department to map side effects and psychosocial challenges related to breast cancer treatment.
Research Group at SUS
The PBCB research group at Stavanger University Hospital is an interdisciplinary research group consisting of research coordinators, researchers, and clinicians from the Department of Surgery, Department of Pathology, Department of Blood and Cancer Diseases, and the Research Department. The research group also includes user representatives. The group was founded by professor and surgeon Håvard Søiland, but is currently led by chief engineer and researcher Kjersti Tjensvoll.
At the Department of Pathology, several retrospective studies have been conducted based on previously archived tumor tissue collected from breast cancer patients between 1989-2004. These studies combine classical pathology, molecular biology, quantitative immunohistochemistry, and digital pathology. The overarching goal of the studies is to improve current breast cancer diagnostics.
Section for Molecular Quantitative Pathology - Helse Stavanger HF
At the Department of Surgery, the breast and endocrine surgical section is a clinical section that operates on 350 breast cancer patients annually. In recent years, the section has participated in several clinical studies focusing on surgical methods, as well as side effects and complications of treatment. The section is responsible for the inclusion and follow-up of patients participating in PBCB at SUS and currently has a PhD student researching endocrine treatment for estrogen-sensitive breast cancer in patients included in PBCB.
Breast and Endocrine Surgical Outpatient Clinic - Helse Stavanger HF
At the Department of Blood and Cancer Diseases, breast cancer research has long focused on developing methods to detect disease recurrence earlier than current technology allows. Detection of micrometastases in bone marrow in patients with early-stage breast cancer has long been the research focus here. However, in recent years, the research has shifted more towards investigating the significance of circulating tumor cells (CTCs) and cell-free tumor DNA (ctDNA) from plasma as markers to predict disease recurrence and as a tool for measuring treatment effects.
PBCB has recruited in total 1,455 patients who were treated for breast cancer at Haukeland (HUS) or Stavanger (SUH) University Hospitals between 2011 and 2019. The patients are followed for 11 years after inclusion into the study with annually (HUS) and bi-annually (SUH) data collection.
To participate in the PBCB study, patients must meet the following criteria:
Inclusion criteria:
- Women of all ages (>18 years).
- Women diagnosed preoperatively with breast cancer.
Exclusion criteria:
- patients that were unable to complete Norwegian questionnaires.
- patients with a prior history of malignancy.
- patients under 18 years of age.
Sample collection
From all the 1455 patients included in the PBCB study, blood and urine samples are collected annually. At Stavanger University Hospital (SUH), blood and urine samples are also collected semi-annually. The following samples are taken during the patient visits:
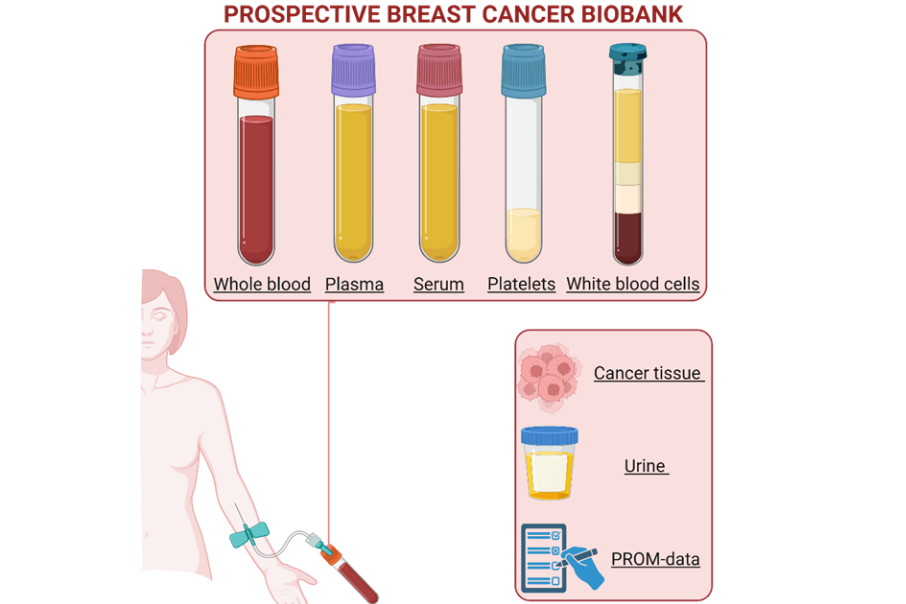
All blood samples collected are processed and stored in different fractions, including whole blood, plasma (warm and cold), serum, platelets, and buffy coat (white blood cells). The table below shows the processing of blood and urine samples, as well as the number of tubes available in the PBCB biobank from each patient at Haukeland and Stavanger University Hospitals.
|
Type of tubes
|
Number of tubes
|
Respite
|
Centrifugation
|
Alliquotation
|
Volume to be distributed
|
Storage
|
|
Vacuette Serum without gel, 6 mL |
8 |
30 -120 min |
2200xg for 10 min at RT |
8 x 0.7 mL, 6 x 0,5 mL |
525 uL serum |
-80°C |
|
4 x 1,0 mL |
865 uL serum |
|||||
|
Vacuette EDTA, 3 mL
|
1 |
|
|
2 x 1,0 mL |
865 uL full blood |
|
|
Vacuette EDTA, 6 mL |
5 |
Max 2 hours |
2200xg for10 min at RT |
6 x 0,7 mL |
525 uL plasma |
|
|
2 x 1,0 mL |
864 uL plasma |
|||||
|
2 x 2,0 mL |
1800 uL plasma |
|||||
|
1 x 1,0 mL |
865 uL buffycoat |
|||||
|
PAXgene, 2,5 mL |
1 |
3-72 hours at RT |
|
Intermediate storage at -20°C for 24 hours, then -80°C. |
||
|
Urine, 20 mL |
1 |
|
|
5 x 2 mL |
1800 uL urine |
-80°C |
|
Type of tubes |
Number of tubes |
Respite |
Centrifugation |
Alliquotation |
Volume to be distributed |
Storage |
|
Vacuette Serum without gel, 10 mL |
2 |
30 -120 min |
1500xg for 25 min at RT |
6 x 0,5 mL |
3 mL serum |
-80°C |
|
4 x 2 mL |
6 mL serum |
|||||
|
Vacuette EDTA, 6 mL
|
1 |
|
|
2 x 2 mL |
3 mL fulblood |
|
|
Vacuette EDTA, 9 mL |
5 |
Max 2 hours |
2 tubes, 2200xg for 10 min at RT |
6 x 2 mL |
12 mL plasma (varm) |
|
|
1 tube (on ice), 2500xg at 4°C |
6 x 0,5 mL |
3 mL plasma (cold) |
||||
|
2 x 1 mL |
2 mL plasma (cold) |
|||||
|
1 x 0,5 mL |
0,5 mL buffycoat |
|||||
|
1 tube (on ice), i.) 200xg for 20 min without brake at 4°C, ii.) 200xg for 10 min without brake at 4°C, iii.)1000xg for 10 min at 4°C |
1 x 0,5 mL |
0,5 mL blood platelets |
||||
|
CPT, 8 mL |
1 |
|
i) 1500xg for 25 min at RT ii) 1000xg for 10 min |
|
|
|
|
PAXgene, 2,5 mL |
1 |
3-72 hours at RT |
|
|
|
Intermediate storage at -20°C for 24 hours, then -80°C. |
|
Urine, 20 mL |
1 |
|
|
6 x 2 mL |
9 mL urine |
-80°C |
RT: Room temperature
|
Name |
Description |
|
EORTC-QLQ-C30 EORTC-QLQ-BR23 |
Quality of life questionnaire. The questionnaires consist of 53 questions about daily activities, physical and mental. |
|
The Mishel Uncertainty in Illness Scale (MUIS) |
4 statements that map how information about illness and treatment has been perceived/understood |
|
Hospital Anxiety and Depression Scale (HAD) |
14 questions of which 7 assess anxiety and 7 depression. |
|
Functional Assessment of Cancer Therapy-Fatigue (FACT-F) |
13 questions about fatigue in the past 7 days |
|
Functional Assessment of Cancer Therapy-Endocrine Scale (FACT-ES) |
56 questions about how patients have felt in the last 7 days. The questions include physical and mental conditions as well as side effects of endocrine treatment. |
|
Functional Assessment of Cancer Therapy-Breast (FACT-B) |
10 questions about quality of life in breast cancer patients |
|
Fatigue Severity Scale (FSS) |
9 questions about how one has experienced feelings of tiredness and exhaustion in the past week. |
|
fatigue Visual Analog Skala (fVAS) |
VAS is a line where on the far left of the line it is 0 = "no problems with tiredness and exhaustion" and on the far right is 100 = "as much tiredness and exhaustion as it is possible to have. |
|
Food Form |
Questionnaire avoiding any of 36 different foods with response alternatives "Yes", "No", "Partial". Seven additional questions about the use of milk and milk products, and one about the use of alcohol. |
|
Questions about joint pain |
3 questions about joint pain with answer option yes/no and grading of morning stiffness in the joints and any muscle pain. |
|
Compliance |
A modified version of the Morisky-8 item Medication Adherence Scale (MMAS) which also contains 2 statements about "belief in the drug" |
|
Subjektive Helseplager (Subjective Health Complaints, SHC). |
The questionnaire consists of 29 questions about subjective, somatic and psychosocial problems during the last 30 days. The SHC has four subgroups: musculoskeletal pain, pseudoneurology, gastrointestinal distress, allergies and colds |
|
Second |
Questions about side effects from the use of tamoxifen and aromatase inhibitors. |
Consent and Participants' Rights
Providing biological material to PBCB is voluntary and requires consent. It will not affect treatment if one chooses not to provide a sample, or if one later wishes to withdraw. Relevant participants for the biobank will be asked during a consultation at the hospital if they wish to contribute biological material and data to PBCB. Participants receive verbal information about the biobank, as well as an information letter. Participants consent to the material and data being used for future research on breast cancer. New projects that will use material and data from PBCB will, however, be evaluated by a regional committee for medical and health research ethics (REK). Such a committee will approve the use of samples/data in individual studies if they believe that the study is covered by the consent given by the participant.
The biological material and data collected in PBCB will be stored permanently in the biobank. All other information will be linked to specified research projects. Such information will be deleted/anonymized when the various projects are completed. It will not be possible to identify participants in the results of the various studies when these are published. Participants can withdraw their consent at any time without it affecting the treatment provided at the hospital. If the participant withdraws from the study, they can choose whether already collected biological material can be used for further research or if it should be destroyed.
User Involvement in the Project
PBCB has an active collaboration with two user representatives from the regional breast cancer association. Both serve as active partners in the User Involvement program, and participate in our project meetings. They are both former breast cancer patients and have provided valuable input on several aspects of the PBCB projects. This has given an important boost to the study group.
At the request of the users, PBCB has among other things started a new research area: Fatigue and workplace participation among breast cancer survivors. Furthermore, our users are actively involved in the design of consent forms and information letters sent to patients. They also provide important input on applications for research funding as well as useful advice when results are to be communicated in various media (written and oral).
In PBCB, a steering committee has been appointed to ensure that the biological material and associated data are managed in accordance with applicable laws, regulations, and internal instructions for research. The steering committee is thus responsible for safeguarding the interests of the participants. It therefore processes applications for new research projects that wish to access the collected biological material or access results from analyses. The steering committee also facilitates collaboration with external institutions to the extent that the activities in PBCB warrant it.
The steering committee consists of the following members:
|
Role |
Name |
Position |
|
Chair |
Prof. Gunnar Mellgren |
Clinical Director of the Laboratory Clinic, HUS. |
|
Board Member |
Kjersti Tjensvoll, PhD |
Chief Engineer at the Department of Blood and Cancer Diseases, and Head of PBCB at SUS. |
|
Board Member |
Kristin Jonsdottir, PhD |
Advisor and Biobank Coordinator, SUS |
|
Board Member |
Thomas Helland, PhD |
Researcher and Biobank Coordinator, HUS |
|
Board Member |
Tone Hoel Lende, PhD |
Department Head at the Department of Surgery and Head of the Section for Breast and Endocrine Surgery, SUS. |
|
Board Member |
Prof. Emiel Janssen |
Head of Research at the Department of Pathology, SUS. |
|
Board Member |
Ann Cathrine Kroksveen, PhD. |
Head of Biobank Haukeland, HUS. |
|
Board Member |
Kristin Viste, PhD |
Senior Physician, Hormone Laboratory |
|
Board Member |
Anette Heie, PhD |
Head, Department of Breast and Endocrine Surgery, Haukeland University Hospital |
- Folke Hermansen Foundation
- Helse Vest
- Inge Steensland Foundation
- Sparebank 1 Foundation: Breast Cancer Research at Helse Stavanger HF – Sparebankstiftelsen SR-Bank
- National Network for Breast Cancer Research
- Internal Research Fund at HUS and SUS
- Aftenbladet 06.10.2013: SUS-leger jobber på fritiden med å løse kreftgåten
- NRK.no 13.06.2020: Fann teikn på kreft i blodprøve lenge før det blei synleg på røntgen – NRK Rogaland – Lokale nyheter, TV og radio
- Rogalands avis 23. juni 2020: Gjennombrudd i brystkreftforskningen ved SUS (for abonnenter)
- Artikkel i Dagens Medisin 09.02.2022: https://www.dagensmedisin.no/debatt-og-kronikk/ja-til-landsomfattende-klinisk-studie-med-lang-oppfolging/224660
- Dagsavisen 26.08.2022: Han får årets forskningspris – Dagsavisen
- Helse Stavanger 26.08.2022: Håvard Søiland får årets forskningspris - Helse Stavanger HF
- Helse Stavanger 26.08.2022: Ti millioner til brystkreftforskning - Helse Stavanger HF
- Jærbladet 17.12.2023: Helse, Hå | Forskinga hans kan vera skilnaden på liv og død. Nå får han heimkommunen med seg (for abonnenter)
- Podcast 26.02.2024: Forskning på brystkreft, tilbakefall og Jærens gærneste mann med Håvard Søiland - Femihelse får barn | Podme
- Dagsavisen 12.01.2024: Vil slå brystkreften sjakk matt med forskningsprosjekt – Dagsavisen
For participants:
- Research Coordinator Stavanger,
email: pbcb@sus.no or phone: 51514434
- Research Coordinator Bergen: Solveig Johanne Skjoldal,
email: solveig.johanne.skjoldal@helse-bergen.no
For researchers:
- Regional leader of PBCB: Professor Gunnar Mellgren,
email: gunnar.mellgren@helse-bergen.no
- Leader of PBCB Stavanger: Kjersti Tjensvoll,
email: kjersti.tjensvoll@sus.no
- Biobank coordinator Stavanger: Kristin Jonsdottir,
email: kristin.jonsdottir@sus.no
- Biobank leader Biobank Haukeland: Ann Cathrine Kroksveen,
email: ann.cathrine.kroksveen@helse-bergen.no
Personalized Tamoxifen Therapy
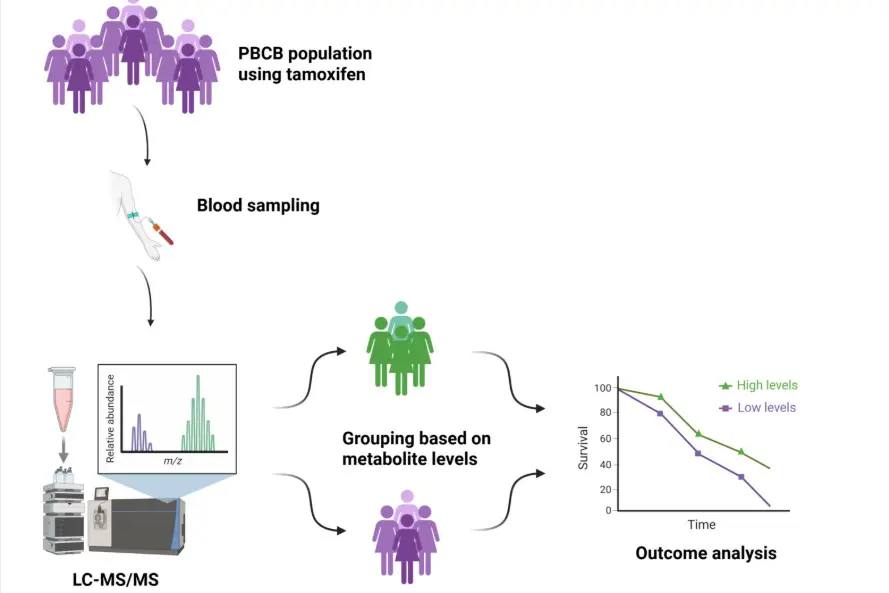
Approximately three-quarters of all breast cancer cases involve estrogen receptor–positive tumors. These patients typically receive adjuvant anti-estrogen therapy for 5–10 years to reduce the risk of recurrence. Tamoxifen plays a central role in anti-estrogen treatment for breast cancer, particularly among premenopausal patients.
Individual genetically determined hepatic metabolism of tamoxifen results in substantial variation in serum concentrations of its active metabolites. These metabolites are approximately 100 times more effective at inhibiting cell division in any residual breast cancer cells following surgery. Dr. Thomas Helland has published two studies showing that patients with low levels of active tamoxifen metabolites may have poorer long-term survival compared to those with higher levels. The PBCB material will be used to validate these findings once sufficient follow-up time has been achieved. Such measurements could potentially provide clinicians with a tool to optimize endocrine adjuvant therapy, for example by tailoring the dosage and type of anti-estrogen treatment to individual patients.
The impact of adherence on clinical outcome in breast cancer patients receiving adjuvant endocrine therapy
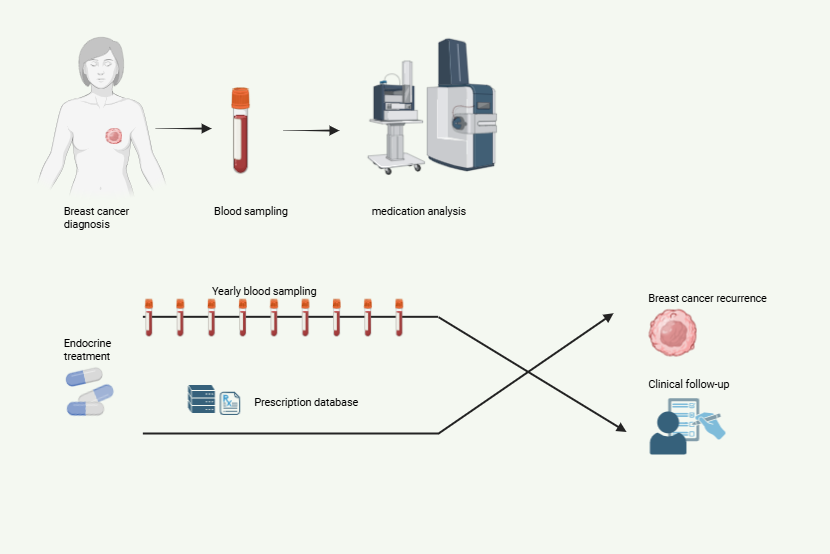
Around 70 % of breast cancer patients have tumors that depend on estrogen for growth and survival. As a result, anti-estrogen therapy is a critical and highly effective adjuvant treatment for these women This therapy typically involves taking an estrogen-blocking tablet daily for up to 10 years, and because it is administered outside of clinical settings, adherence depends entirely on the patient. However, anti-estrogen treatment often causes a range of side effects that can significantly reduce quality of life, increasing the risk of premature discontinuation. Stopping therapy early substantially raises the likelihood of breast cancer recurrence. Identifying patients at high risk of discontinuing anti-estrogen therapy—also known as endocrine therapy—could enable more targeted follow-up and improve long-term survival outcomes in this population.
In this project, PhD candidate Finn Magnus Eliassen investigates whether poor adherence to endocrine therapy leads to higher recurrence rates and increased breast cancer mortality. Data from prescription registries are used to evaluate treatment adherence and are compared with analyses of blood samples collected during the same timeframe. The extended follow-up period in PBCB makes it also possible to account for late recurrences, which remain a significant challenge in estrogen-sensitive breast cancer.
In addition, we will also use registered Patient-Reported Outcome Measures (PROM) data to investigate how endocrine therapy influences patients’ self-reported sexual health, and specifically whether poorer sexual health within this group contributes to reduced treatment adherence.
Personalized Monitoring in Breast Cancer (PerMoBreCan)
In the PerMoBreCan project, the aim is to investigate the clinical utility of novel circulating biomarkers (circulating tumor cells, circulating tumor DNA, metabolites, extracellular vesicles, proteins, glycoproteins, and cytokines from blood) for the early detection of systemic relapse in breast cancer patients, enabling timely initiation of new treatment. The project is organized into several sub-studies, which include examination of the tumor itself as well as the new circulating tumor markers in blood samples from breast cancer patients enrolled in the PBCB project.
Sub-project 1: Analysis of breast cancer tumor characteristics and circulating microRNAs derived from extracellular vesicles
In this project, RNA from breast cancer tumors has been sequenced to identify novel microRNA profiles capable of differentiating tumors that are sensitive to adjuvant chemotherapy from those that exhibit resistance. Additionally, tumors will be examined for genetic alterations, with particular focus on mutations in the estrogen receptor gene and other well-characterized oncogenes. The Ki67-Adjusted Mitotic Score (KAMS), which provides an estimate of cell cycle kinetics within the proliferative cell population, will also be evaluated. Centrosome amplification, serving as an indicator of tumor heterogeneity and chromosomal instability, will be analyzed. Additionally, the genomic alterations identified in breast cancer tumors have been mapped and integrated with analyses of circulating tumor DNA to detect cancer-derived DNA in blood, a biomarker indicative of systemic disease.
Moreover, circulating microRNAs derived from extracellular vesicles will be analyzed. MicroRNA profiles associated with chemotherapy-sensitive tumors will be systematically compared to those derived from treatment-resistant tumors. Candidate microRNA markers identified in tumor tissue and correlated with therapeutic response will be further assessed in total RNA isolated from extracellular vesicles obtained from high-risk patients. This strategy aims to determine the potential utility of these microRNAs as biomarkers for treatment monitoring, longitudinal follow-up, and early detection of systemic relapse. In addition, expression levels of selected genes in platelets will be assessed. Bioinformatics analyses are performed by Marie Austdal at the Department of Pathology, Stavanger University Hospital.
Sub-project 2: Validation of novel biomarkers for early relapse in Breast Cancer Patients
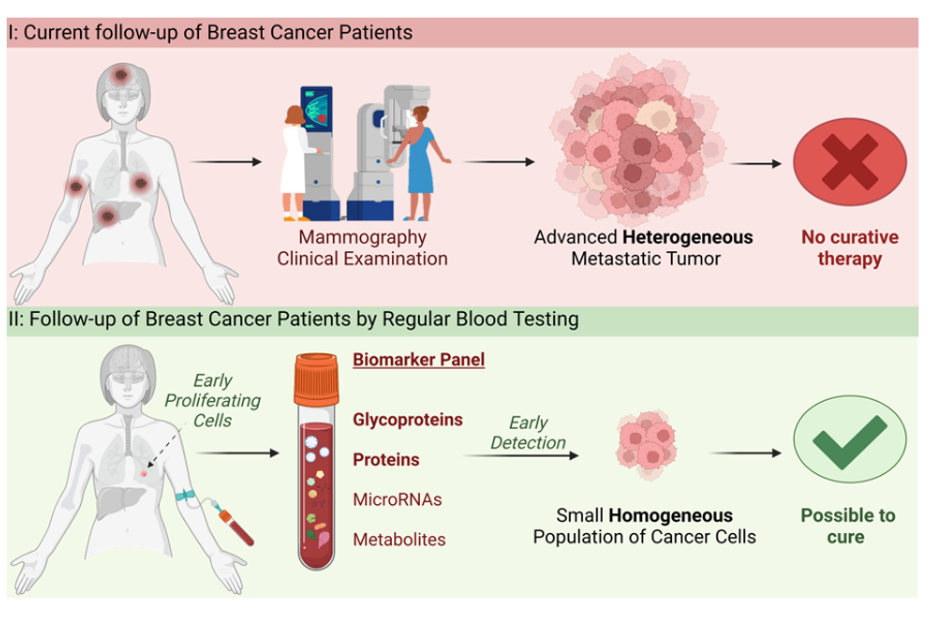
One of the major challenges in breast cancer is the persistent risk of recurrence, which may occur even years after primary treatment. The most critical concern is systemic relapse, characterized by metastatic spreading to distant organs. At present, reliable methods for early detection of systemic disease are lacking; recurrence is typically identified only when a new lesion becomes symptomatic or detectable through radiological imaging. At this advanced stage, curative treatment is rarely feasible, and patients must transition to palliative, life-prolonging therapy.
To enhance patient safety and outcomes, there is an urgent need for diagnostic strategies capable of detecting relapse well before conventional imaging. In this project, we conduct in-depth analyses of blood samples to identify proteins, glycoproteins, metabolites, and microRNAs. By comparing samples from patients with and without relapse, as well as healthy controls, we aim to identify biomarkers that can reveal early signs of recurrence and enable closer, more personalized follow-up. This work is conducted by PhD candidate Marie Landa Austbø at the Department of Pathology, Stavanger University Hospital.
Sub-project 3: Liquid biopsies for personalized follow-up of operable breast cancer patients
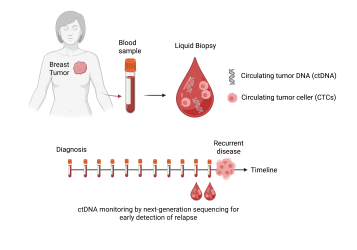
Circulating tumor cells (CTCs) in blood have been shown to be significant for the prognosis of breast cancer patients. The conventional method for detecting these cancer cells (CellSearch) relies on the surface protein EpCAM, which is downregulated in some circulating tumor cells due to an epithelial-to-mesenchymal transition. In this study, we aim to investigate the value of detecting all types of cancer cells in the blood, regardless of whether they express the EpCAM surface protein, in early-stage breast cancer.
The tumor cells are enriched using the immunomagnetic method MINDEC, developed in-house, and subsequently identified through quantitative RT-PCR by measuring mRNA markers that are virtually absent in normal blood cells but expressed in cancer cells. Preoperative blood samples are analyzed to assess their prognostic potential, while semi-annual postoperative samples constitute a unique resource for evaluating the role of these assays in disease surveillance.
Circulating tumor DNA (ctDNA) is another biomarker with considerable potential for prognostication and monitoring disease progression in breast cancer. ctDNA originates from apoptotic or necrotic tumor cells, including those from primary tumors and metastatic sites, and can be detected through tumor-specific mutations. In this study, ctDNA will be identified and characterized using next-generation sequencing (NGS) in plasma samples collected preoperatively and at semi-annual follow-up intervals from PBCB participants. Quantitative changes in ctDNA, reflected by the emergence or increase of specific mutations, will be correlated with radiological findings for comprehensive disease monitoring. This approach will allow us to determine whether ctDNA measurement has clinical significance and to identify potential resistance mutations if patients undergoing chemotherapy. The work is carried out by PhD student Kristin Løge Aanestad from the Cancer and Medical Physics Research Group, Micrometastasis Group, Stavanger University Hospital.
Fatigue in breast cancer survivors
Advances in treatment modalities and early detection have significantly improved breast cancer survival rates; however, many survivors experience long-term sequelae of treatment. Up to 30–40% of women are affected by fatigue, which often impairs their ability to return to work. Despite the considerable burden on both patients and society, knowledge regarding the relationship between breast cancer, its treatment, and such late effects remains limited. Furthermore, the mechanisms underlying why fatigue severely affects some individuals while others remain unaffected are poorly understood.
The PBCB material is particularly well suited for studies investigating mechanisms of persistent fatigue, as it enables a combination of longitudinal clinical, biological, and patient-reported outcome (PROM) data.
REsearch on BrEast Cancer induced chronic conditions supported by Causal Analysis of multi-source data (REBECCA)
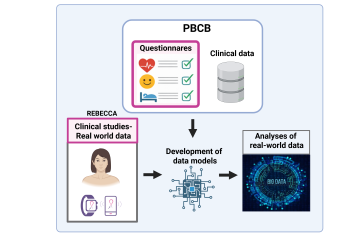
Cancer-related fatigue is among the most prevalent long-term adverse effects following breast cancer treatment and may persist for several years in otherwise healthy survivors. Fatigue constitutes a complex, multidimensional condition that affects patients physically, cognitively, and emotionally, and consequently, for some individuals, exerts a substantial impact on overall functional capacity, quality of life, and occupational performance. Survivors experiencing fatigue may therefore encounter increased financial strain and psychological stress, as well as a higher utilization of healthcare services.
Currently, healthcare professionals primarily assess patients’ quality of life through patient-reported outcome measures (PROMs). These instruments rely on subjective self-assessments, wherein perceptions of fatigue can vary considerably. To address this limitation, Stavanger University Hospital (SUS) participated as the sole Norwegian institution in an EU-funded initiative aimed at characterizing quality of life among breast cancer survivors affected by late effects, including cancer-related fatigue, through the collection of objective lifestyle data. So-called “real-world data” were obtained via smartwatches and the REBECCA mobile application in three clinical REBECCA studies conducted at SUS. In addition, questionnaires and clinical data collected within the PBCB framework were analyzed to explore associations between various parameters and fatigue status (results not yet published). These insights provide a foundation for the development of data-driven models to analyze “real-world data” generated through the REBECCA project. The figure below illustrates an overview of the REBECCA EU project, including the integration of PBCB data analyses. Home - REBECCA
Estrogen levels during long-term treatment with aromatase inhibitors
Aromatase inhibitors are among the most widely used anticancer drugs in the world and play a central role in the treatment of estrogen receptor–positive breast cancer in postmenopausal women. These medications act by blocking the enzyme aromatase, which converts androgens into estrogens in body tissues, particularly in adipose tissue.
Several studies have reported detectable estrogen levels in some patients during therapy. This has raised concerns about whether aromatase inhibitors always provide sufficient estrogen suppression, especially in women with a high body mass index. However, previous studies have been limited by short follow‑up periods, lack of verification of drug exposure, and the use of analytical methods with insufficient sensitivity.
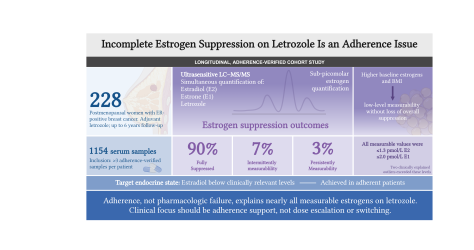
In this project, we used the PBCB cohort to follow 228 postmenopausal women treated with letrozole, with a total of 1,154 serum samples collected over up to six years. Using ultrasensitive mass‑spectrometric assays, we measured estradiol, estrone, and letrozole, where letrozole concentrations provided objective verification of treatment adherence.
The study demonstrates that letrozole induces robust and sustained estrogen suppression. Ninety percent of patients exhibited complete suppression at all time points, and only a small minority had persistently measurable estrogen levels. No biochemical “escape” or gradual increase in estrogen levels over time was observed. Women with higher body mass index and elevated pretreatment estrogen levels more frequently showed low‑level measurable values, but without evidence of clinically meaningful reduction in suppression.
Overall, the findings indicate that incomplete estrogen suppression largely reflects reduced adherence rather than inadequate pharmacologic effect. Clinical follow‑up should therefore prioritize verification and support of treatment adherence, rather than dose escalation or switching endocrine therapy.
PROTAM: Endocrine effects of Tamoxifen treatment
Tamoxifen is an endocrine therapy with a complex and tissue‑specific activity profile in breast cancer treatment. The drug acts primarily by blocking the estrogen receptor in breast tissue but exerts both agonistic and antagonistic effects in other tissues. Despite its extensive clinical use, the systemic hormonal effects of tamoxifen remain insufficiently characterized.
Several studies have reported so‑called off‑target effects of tamoxifen, and recent research has highlighted the importance of interactions among estrogen, progesterone, and androgen receptors in breast cancer biology. Previous investigations of hormonal changes during tamoxifen therapy have, however, been limited by small patient cohorts, analytical methods with inadequate sensitivity, and analyses performed mainly at the group level.
PROTAM (Steroid PROfiling of TAMoxifen) is a research project examining how tamoxifen treatment affects the systemic steroid hormone milieu in both pre‑ and postmenopausal breast cancer patients. By analyzing serum samples collected before and during treatment, we map individual hormonal changes and effects on key endocrine axes over time. The samples are analyzed using mass‑spectrometric methods that collectively cover large parts of steroidogenesis and enable detailed hormonal profiling at a level previously not achievable. To date, 350 serum samples from the PBCB cohort have been prepared for analysis.
The results may provide new insights into the systemic endocrine effects of tamoxifen treatment and help elucidate hormonal changes relevant to adverse effects and individual variation in treatment response.
|
PhD kandidat |
Veiledere |
Tittel |
Status |
|
Thomas Helland, molekylærbiolog |
Gunnar Mellgren, Håvard Søiland, Emiel Janssen |
Tamoxifen i behandling av luminal brystkreft - Implikasjoner av aktive metabolitter for genuttrykk, bivirkninger og klinisk utfall. |
Disputert 22.02.2019 |
|
Kari Britt Hagen, sykepleier |
Ragna Lind, Håvard Søiland |
Brystkreft: Etterlevelse av adjuvant endokrin behandling hos pasienter i Vest-Norge, |
Disputert 23.09.2019 |
|
Finn Magnus Eliassen, overlege |
Gunnar Mellgren, Tone Hoel Lende, Thomas Helland |
Betydning av adherence for klinisk utfall hos pasienter med brystkreft som får adjuvant endokrin behandling |
PhD prosjekt startet |
|
Marie Austdal, molekylærbiolog |
Emiel Janssen |
Persontilpasset monitorering av brystkreft (PerMoBreCan) |
Post doc. prosjekt ferdig |
|
Marie Landa Austbø, molekylærbiolog |
Emiel Janssen |
Validering av nye biomarkører for påvisning av tidlig tilbakefall hos brystkreftpasienter |
PhD prosjekt startet |
|
Kristin Løge Aanestad, lege |
Bjørnar Gilje, Oddmund Nordgård og Kjersti Tjensvoll |
Flytende biopsier for persontilpasset oppfølging av pasienter med opererbar brystkreft
|
PhD prosjekt startet |
- Monitoring of circulating tumor DNA allows early detection of disease relapse in patients with operable breast cancer. Mol Oncol. 2025 Nov 27. doi: 10.1002/1878-0261.70170. Online ahead of print.
- Quantitative proteomics reveals serum proteome alterations during metastatic disease progression in breast cancer patients. Clin Proteomics. 2024 Jul 29;21(1):52. doi: 10.1186/s12014-024-09496-3.
- Importance of endocrine treatment adherence and persistence in breast cancer survivorship: a systematic review. BMC Cancer. 2023 Jul 4;23(1):625.
- Simultaneous Quantification of Aromatase Inhibitors and Estrogens in Postmenopausal Breast Cancer Patients. The Journal of Clinical Endocrinology & Metabolism, Volume 107, Issue 5, May 2022, Pages 1368–1374.
- Liquid biopsies and patient-reported outcome measures for integrative monitoring of patients with e…. BMJ Open. 2022 Apr 29;12(4):e054404.
- Simultaneous Quantification of Aromatase Inhibitors and Estrogens in Postmenopausal Breast Cancer P…. J Clin Endocrinol Metab. 2021 Dec 27;107(5):1368–1374.
- Effect of Genetic Variability in 20 Pharmacogenes on Concentrations of Tamoxifen and Its Metabolites. J Pers Med. 2021 Jun 4;11(6):507.
- Drug monitoring of tamoxifen metabolites predicts vaginal dryness and verifies a low discontinuatio…. Breast Cancer Res Treat. 2019 Aug;177(1):185-195. doi: 10.1007/s10549-019-05294-w. Epub 2019 May 29.
- Adherence to adjuvant endocrine therapy in postmenopausal breast cancer patients: A 5-year prospect…. Breast. 2019 Apr:44:52-58. doi: 10.1016/j.breast.2019.01.003. Epub 2019 Jan 8.
- Influence of pre-operative oral carbohydrate loading vs. standard fasting on tumor proliferation and clinical outcome in breast cancer patients ─ a randomized trial. BMC Cancer. 2019 Nov 8;19(1):1076.
- Metabolic consequences of perioperative oral carbohydrates in breast cancer patients - an explorative study. BMC Cancer. 2019 Dec 4;19(1):1183.
- Diet in women with breast cancer compared to healthy controls - What is the difference? Eur J Oncol Nurs. 2018 Feb:32:20-24. doi: 10.1016/j.ejon.2017.11.003. Epub 2017 Nov 24.
- Fatigue, anxiety and depression overrule the role of oncological treatment in predicting self-repor…- Breast. 2016 Aug:28:100-6. doi: 10.1016/j.breast.2016.05.005. Epub 2016 Jun 2.
- In patients younger than age 55 years with lymph node-negative breast cancer, proliferation by mitotic activity index is prognostically superior to adjuvant! J Clin Oncol. 2011 Mar 1;29(7):852-8. doi: 10.1200/JCO.2009.25.0407. Epub 2010 Dec 28.