Research projects
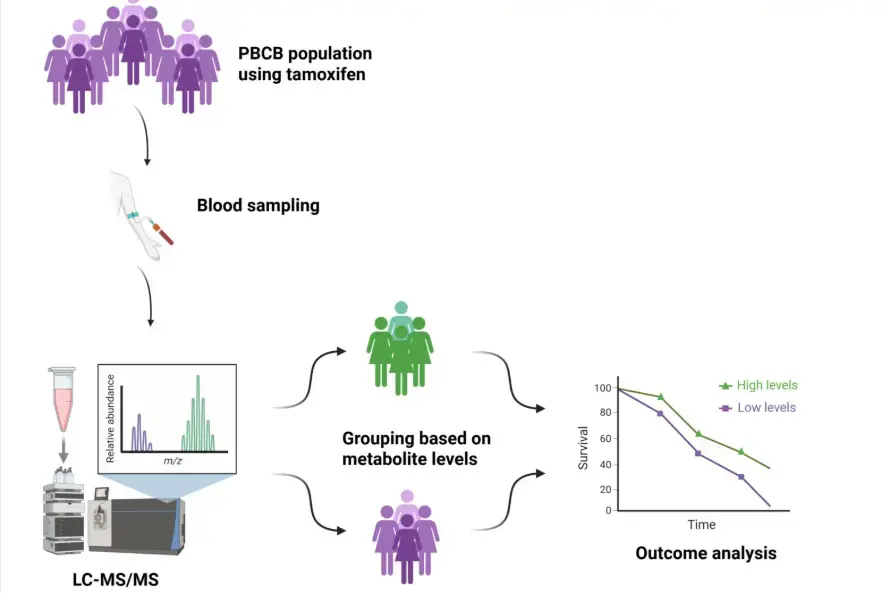
Patients with oestrogen receptor-positive tumours account for approximately three-quarters of all breast cancer cases. In these patients, oestrogen receptors are found in the cancerous tissue, which can be exploited in treatment (so-called endocrine adjuvant therapy). Patients then receive anti-oestrogen adjunct treatment for 5 to 10 years to reduce the risk of recurrence. Tamoxifen is a medication that plays a central role in such anti-oestrogen treatment, especially for younger and premenopausal patients.
Tamoxifen is metabolised differently among patients, and the individual (genetically determined) metabolism of tamoxifen in the liver leads to highly variable serum levels of the active forms of tamoxifen. The active forms (active metabolites) are 100 times more effective at stopping cell division of any remaining breast cancer cells after surgery. Dr Thomas Helland has published two studies indicating that patients with low levels of active tamoxifen metabolites are likely to have poorer long-term survival than patients with higher levels. The PBCB material will be used to validate this finding when the follow-up period is long enough. Such measurements could potentially provide clinicians with a tool to optimise endocrine adjuvant treatment by, for example, administering patients a tailored amount and type of anti-oestrogen.
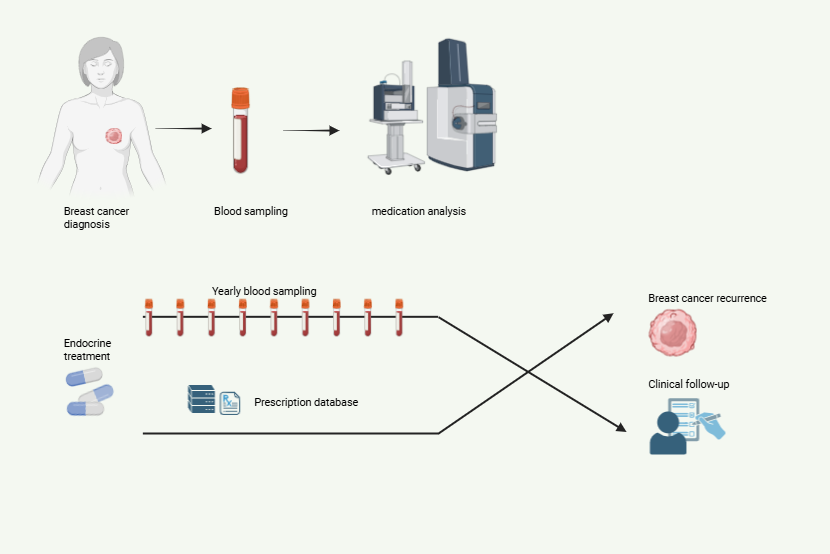
Seventy per cent of breast cancer patients have tumours that have become dependent on oestrogens for survival. Therefore, anti-oestrogen treatment is a highly effective and important adjunct therapy (adjuvant endocrine treatment) for these women. This form of therapy involves taking an oestrogen-blocking tablet daily for up to ten years, and is therefore conducted without supervision from healthcare personnel. Since anti-oestrogen treatment often generates a range of side effects that reduce quality of life, this increases the risk that patients may choose to discontinue treatment far too early. Unfortunately, this results in an increased risk of breast cancer recurrence. Identifying patients at high risk of discontinuing anti-oestrogen treatment could improve follow-up care and enhance long-term survival for these women.
In this project, PhD candidate Finn Magnus Eliassen focuses on whether poor treatment adherence (that is, whether the patient actually takes the medication) to endocrine treatment leads to more recurrences and increased mortality from breast cancer. Prescription register data will be used to assess adherence, and this data will be compared with analyses of blood samples taken during the same period. The long follow-up time in PBCB allows for consideration of late recurrences, which is a particular challenge with oestrogen-sensitive breast cancer.
Furthermore, we will utilise recorded PROM data (responses to questionnaires) to examine how endocrine treatment affects the self-reported sexual health of patients, and specifically whether poorer sexual health in this patient group leads to worse adherence to treatment.
In the PerMoBreCan project, the aim is to investigate the clinical utility of novel circulating biomarkers (such as circulating tumour cells, circulating tumour DNA, metabolites, extracellular vesicles, proteins, glycoproteins and cytokines from blood) for the early detection of potential systemic relapse in breast cancer patients, so that new treatment can be initiated at an early stage. The project is organised into several sub-projects, including analyses of the tumour itself as well as the relevant novel circulating tumour markers in blood samples from breast cancer patients enrolled in the PBCB project.
Sub-project 1: Analysis of breast tumours and circulating microRNA from extracellular vesicles
In this project, RNA from breast tumours has been sequenced to identify novel microRNA profiles that can distinguish between tumours that are sensitive to adjuvant chemotherapy and those that are resistant. In addition, the breast tumours will be examined for genetic alterations, particularly in the oestrogen receptor gene and other known cancer genes (oncogenes). The so-called Ki67-adjusted Mitotic Score (KAMS), which provides a measure of cell cycle kinetics in the proliferative cell population, will also be assessed. Centrosome amplification (as a measure of tumour heterogeneity and chromosomal instability) will be analysed.
Genetic alterations in the breast tumours have also been mapped, and this analysis will be linked to the analysis of circulating tumour DNA in order to identify tumour-derived DNA in the blood as an indicator of cancer dissemination.
In addition, circulating microRNA from extracellular vesicles will be analysed. MicroRNA profiles from breast tumours that are sensitive to adjuvant chemotherapy will be compared with profiles from more treatment-resistant tumours. Relevant microRNA markers (identified through analyses of breast tumours and associated with treatment response) will be analysed in total RNA isolated from extracellular vesicles from high-risk patients. This will allow us to assess whether these microRNAs can be used for treatment monitoring, follow-up, and early detection of systemic relapse. In addition, gene expression of selected genes in platelets will be investigated. The work is carried out by bioinformatician Marie Austdal at the Department of Pathology, Stavanger University Hospital.
Sub-project 2: Validation of novel biomarkers for the detection of early relapse in breast cancer patients
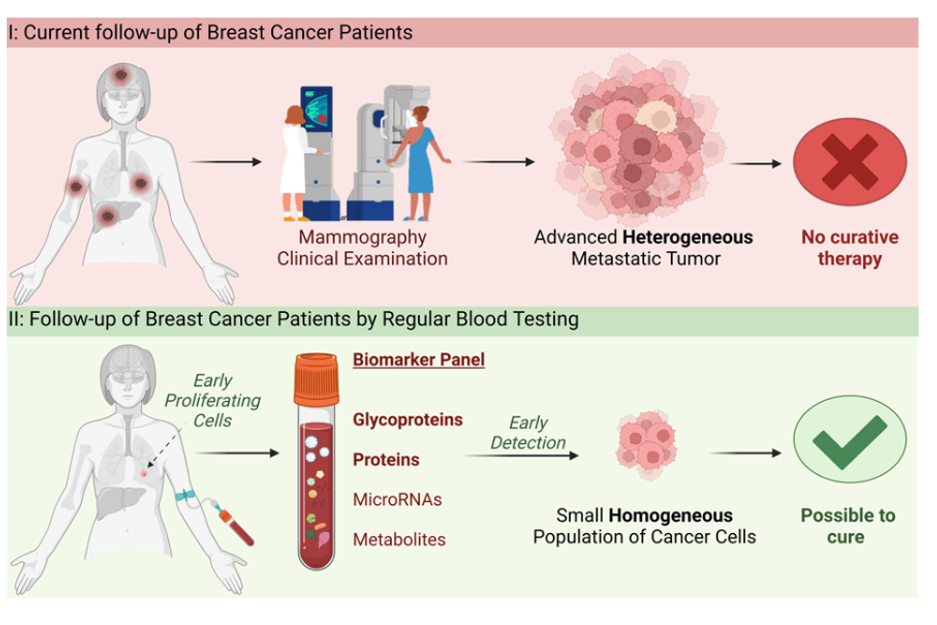
A major challenge in breast cancer is the risk of relapse, which can occur many years after treatment. The most serious concern is systemic relapse, i.e. when the cancer spreads to other organs in the body. At present, we lack effective methods for detecting this at an early stage, and the disease is often only identified when a new tumour causes symptoms or becomes visible on radiological imaging. When relapse is detected this late, curative treatment is usually no longer possible, and the patient must instead transition to life-prolonging therapy.
To provide patients with greater reassurance, there is a need for methods that can detect relapse long before current diagnostic approaches. In our project, we analyse blood samples for proteins, glycoproteins, metabolites and microRNA. By comparing samples from patients with and without relapse, as well as from healthy controls, we aim to identify markers that can reveal early signs of relapse and enable closer and more personalised follow-up. The work is carried out by PhD candidate Marie Landa Austbø at the Department of Pathology, Stavanger University Hospital.
Sub-project 3: Liquid biopsies for personalised follow-up of patients with operable breast cancer
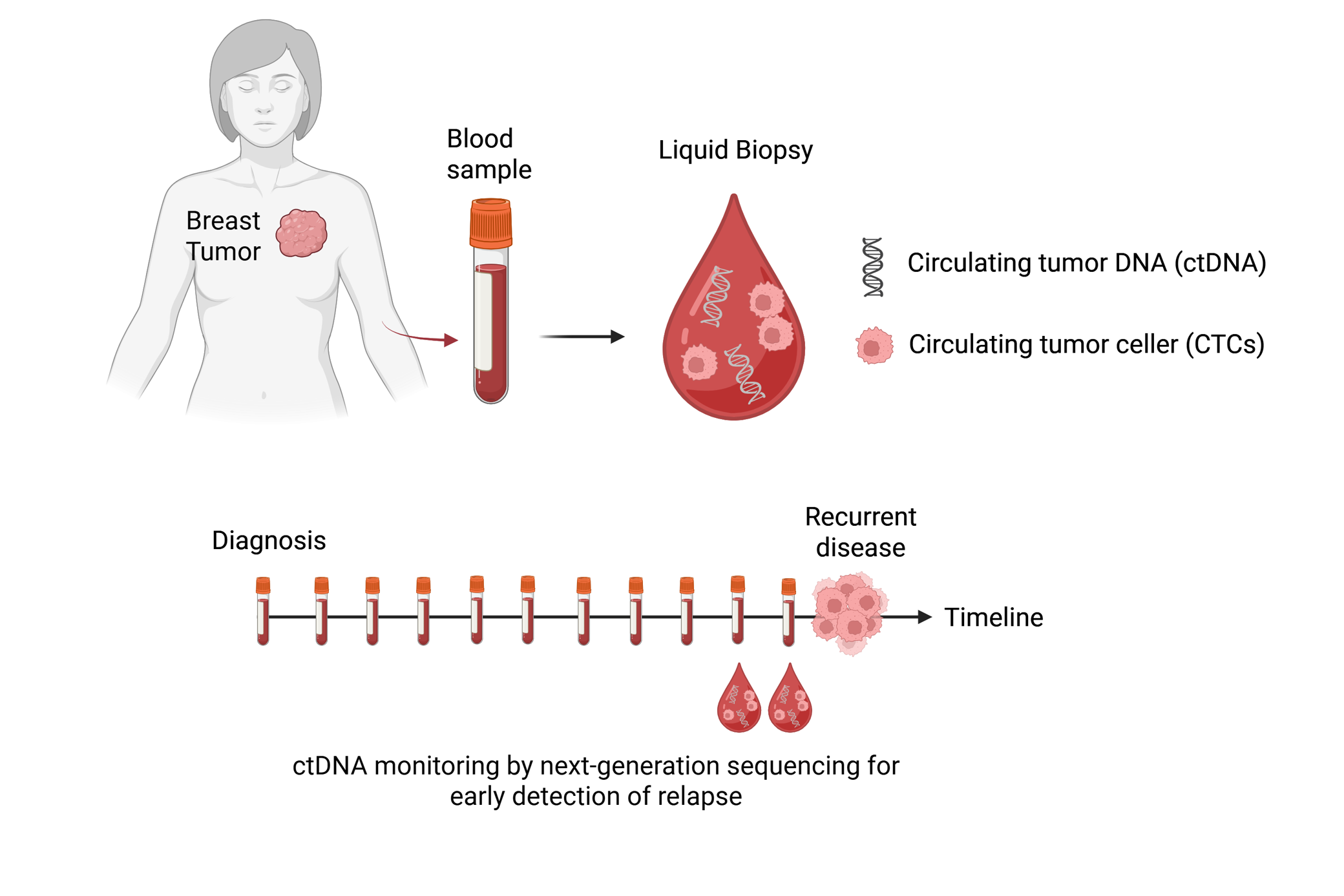
Circulating tumour cells (CTCs) in the blood have been shown to be of prognostic significance in breast cancer patients. The conventional method for detecting these cancer cells (CellSearch) depends on the surface protein EpCAM, which is downregulated in a proportion of circulating tumour cells as a result of epithelial-to-mesenchymal transition. In this study, we will investigate the utility of detecting all types of cancer cells in the blood, regardless of whether they express the EpCAM surface protein, in early-stage breast cancer.
Tumour cells are enriched using the immunomagnetic MINDEC method (developed in-house) and detected by quantitative RT-PCR through measurement of mRNA markers that are largely absent in normal blood cells but present in cancer cells. Blood samples collected prior to surgery are analysed with the aim of predicting prognosis. Semi-annual blood samples collected after surgery constitute a unique resource for evaluating the value of these analyses in disease monitoring.
Circulating tumour DNA (ctDNA) is another promising biomarker for prognosis and disease progression in breast cancer. ctDNA originates from dying cells in the primary breast tumour or from potential metastases and can be detected using tumour-specific mutations. In this study, ctDNA will be detected and characterised using next-generation sequencing (NGS), with plasma samples collected prior to surgery as well as semi-annual follow-up samples from patients in the PBCB study being analysed. Levels of ctDNA, reflected by any increase in the frequency of specific mutations, will be correlated with imaging findings for disease monitoring. In this way, we can both determine whether ctDNA measurement has clinical significance and identify potential resistance mutations if patients receive chemotherapy. The work is carried out by PhD candidate Kristin Løge Aanestad from the Research Group for Cancer and Medical Physics, Micrometastasis Group, Stavanger University Hospital.
Fatigue in breast cancer survivors
New treatment methods and earlier detection mean that more patients survive breast cancer, but many survivors experience long-term side effects from treatment. Up to 30–40 % of women are affected by fatigue (severe or chronic exhaustion), which can make it difficult to return to work. Despite the considerable burden for both patients and society, little is known about the relationship between breast cancer, cancer treatment, and such late effects. The mechanisms underlying why some individuals are severely affected by fatigue while others are not remain poorly understood.
The PBCB dataset is well suited for studies investigating the mechanisms of persistent fatigue, as longitudinal clinical, biological, and patient-reported outcome (PROM) data can be triangulated.
Cancer-related fatigue is one of the most common late effects following breast cancer treatment and can persist for several years in otherwise healthy survivors. Fatigue is a complex, multidimensional condition that affects patients physically, mentally, and emotionally, and therefore has a significant impact on overall functioning, quality of life, and work capacity for some patients. Cancer survivors affected by fatigue may therefore experience increased financial burden and stress, and have a higher consumption of healthcare services.
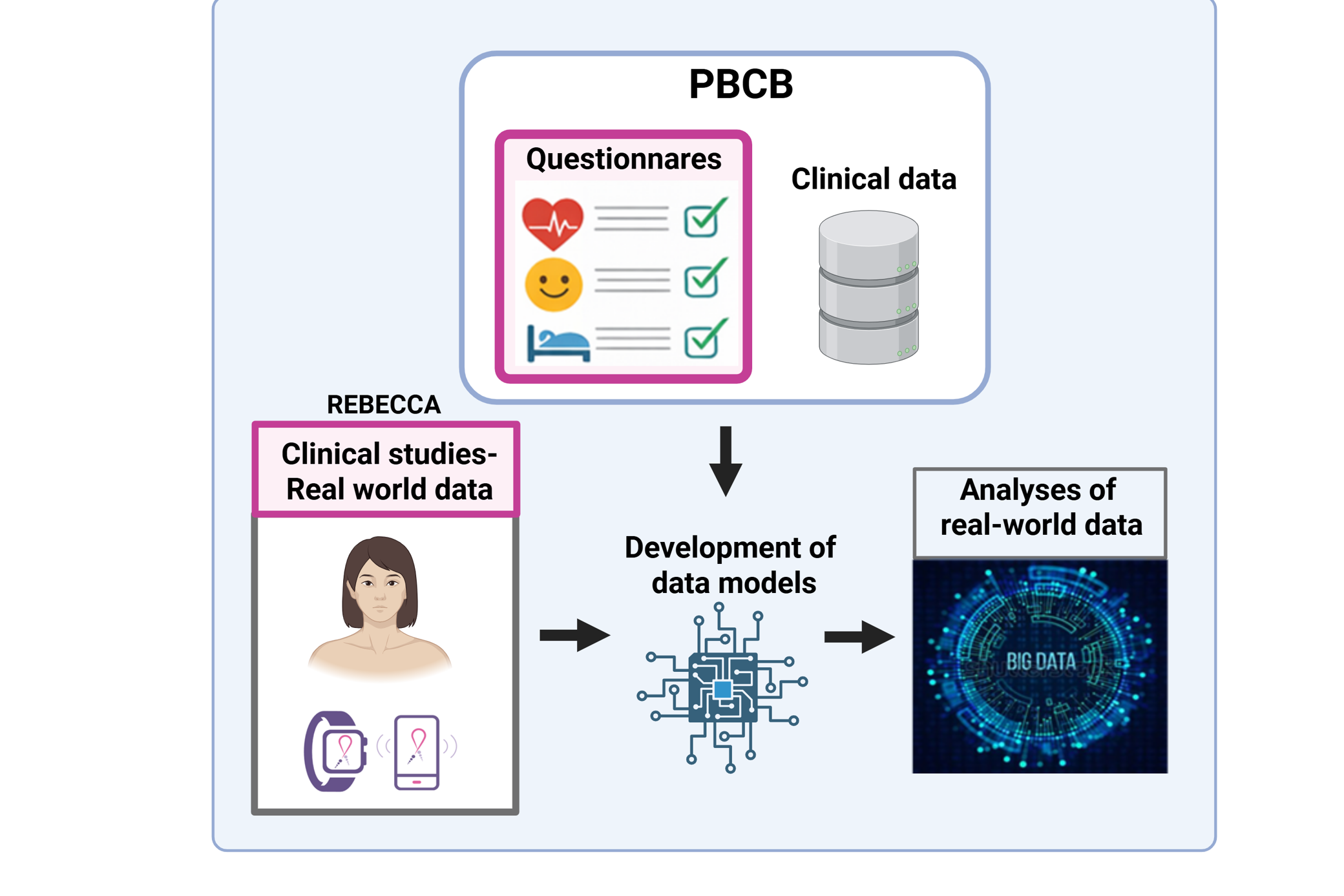
Today, healthcare professionals gain insight into patients' quality of life primarily through questionnaires (PROMs). These questionnaires are based on patients' subjective assessments, where the perception of fatigue can vary significantly. SUS participated as the only Norwegian institution in an EU-funded project aimed at mapping the quality of life of breast cancer survivors affected by late effects, including cancer-related fatigue, through the collection of objective lifestyle data.
So-called “real-world data” was collected using smartwatches and a REBECCA mobile app in three clinical REBECCA studies at SUS. In this context, the questionnaires and clinical data collected in PBCB were also analysed to study the relationships between various parameters and fatigue status (results not published). This insight further forms the basis for the development of data models that analyse “real-world data” from the REBECCA project. The figure above provides an overview of the REBECCA EU project, which includes analyses of the PBCB data.

Aromatase inhibitors are among the most commonly used cancer medications in the world and are central to the treatment of oestrogen receptor-positive breast cancer in postmenopausal women. The drugs work by blocking the enzyme aromatase, which converts androgens to oestrogen in the body, particularly in adipose tissue.
Several studies have reported measurable oestrogen levels in some patients during treatment. This has raised questions about whether aromatase inhibitors always provide sufficient oestrogen suppression, particularly in women with a high body mass index. However, previous studies have had limited follow-up time, lacked verification of drug exposure, and employed analytical methods without sufficient sensitivity.
In this project, we used PBCB to follow 228 postmenopausal women treated with letrozole, with 1154 serum samples collected over up to six years. Using ultra-sensitive mass spectrometric methods, we measured oestradiol, oestrone, and letrozole, where letrozole levels provide an objective verification of treatment adherence.
The study shows that letrozole provides robust and sustained oestrogen suppression. Ninety per cent of patients were fully suppressed at all measurement time points, and only a small minority had persistently measurable oestrogen levels. No biochemical "escape" or gradual increase in oestrogens over time was observed. Women with a high body mass index and higher oestrogen levels before treatment initiation had somewhat more frequent low, measurable values, but without signs of clinically relevant failure in suppression.
Overall, the findings indicate that incomplete oestrogen suppression largely reflects reduced adherence rather than insufficient drug effect. Clinical follow-up should therefore prioritise verification and support of treatment adherence, rather than dose escalation or switching endocrine therapy.
Tamoxifen is an endocrine medication with a complex and tissue-specific action profile in breast cancer treatment. The drug primarily works by blocking the oestrogen receptor in breast tissue, but exerts both agonistic and antagonistic effects in other tissues. Despite extensive clinical use, the impact of tamoxifen on the systemic hormonal environment remains inadequately mapped.
Several studies have reported so-called off-target effects of tamoxifen, and recent research has highlighted the importance of the interplay between oestrogen, progesterone, and androgen receptors in breast cancer biology. However, previous investigations of hormonal changes during tamoxifen treatment have been limited by small patient samples, analysis methods with insufficient sensitivity, and analyses primarily at the group level.
PROTAM (Steroid Profiling of TAMoxifen) is a research project that examines how tamoxifen treatment affects the systemic steroid hormone environment in both pre- and postmenopausal breast cancer patients. By analysing serum samples before and during treatment, we map individual hormonal changes and the influence on central endocrine axes over time. The samples are analysed using mass spectrometric methods that collectively cover large parts of steroid synthesis and enable detailed hormone profiling at a level that has not previously been possible. So far, 350 serum samples from PBCB have been prepared for analysis.
The results may provide new insights into the systemic endocrine effects of tamoxifen treatment and help illuminate hormonal changes relevant to side effects and individual variation in treatment response.
| PhD candidate | Supervisors | Title | Status |
|---|---|---|---|
| Thomas Helland, molekylærbiolog | Gunnar Mellgren, Håvard Søiland, Emiel Janssen | Tamoxifen in the treatment of luminal breast cancer – implications of active metabolites for gene expression, side effects and clinical outcomes | Disputert 22.02.2019 |
| Kari Britt Hagen, sykepleier |
Ragna Lind, Håvard Søiland |
Breast cancer: Adherence to adjuvant endocrine therapy among patients in Western Norway |
Disputert 23.09.2019 |
| Finn Magnus Eliassen, overlege |
Gunnar Mellgren, Tone Hoel Lende, Thomas Helland |
The impact of adherence on clinical outcomes in patients with breast cancer receiving adjuvant endocrine therapy | PhD-prosjekt startet |
| Marie Austdal, molekylærbiolog |
Emiel Janssen |
Personalised monitoring of breast cancer (PerMoBreCan) |
Postdok-prosjekt ferdig |
| Marie Landa Austbø, molekylærbiolog |
Emiel Janssen |
Validation of novel biomarkers for the detection of early relapse in breast cancer patients | PhD-prosjekt startet |
| Kristin Løge Aanestad, lege |
Bjørnar Gilje, Oddmund Nordgård og Kjersti Tjensvoll |
Liquid biopsies for personalised follow-up of patients with operable breast cancer | PhD-prosjekt startet |